The Efficacy of Caldolor
See How Caldolor Can Reduce Pain and Opioid Use in Different Surgical Settings
Demonstrated Fever Reduction in Pediatrics1
Caldolor is as or more efficacious than acetaminophen for the treatment of pain and fever in adult and pediatric populations and is equally safe.52
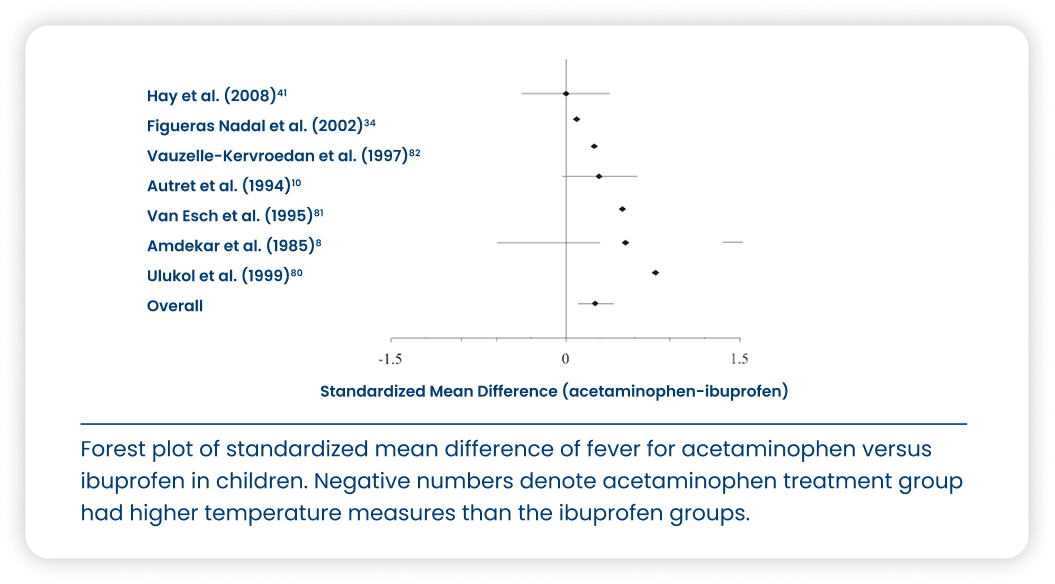
Study description
A meta-analysis of 85 studies that directly compared ibuprofen to acetaminophen; of the 85 studies, 54 contained analgesic efficacy data, 35 contained antipyretic/temperature reduction data, and 66 contained safety data.
Caldolor significantly reduces temperature in febrile patients at 2h & 4h compared to acetaminophen.51
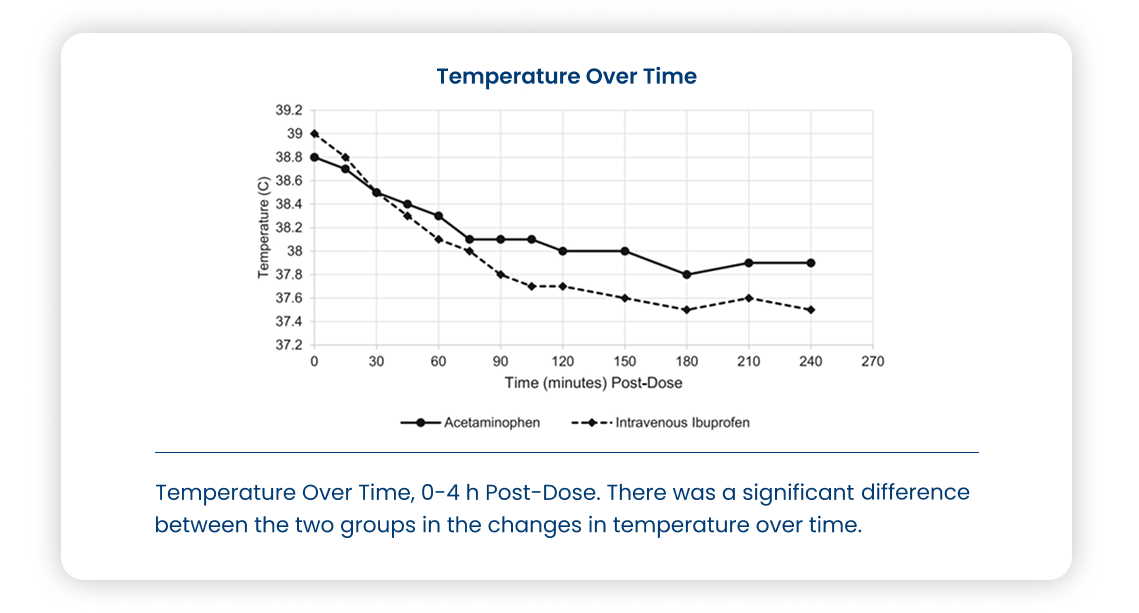
Study description
Multicenter, randomized, open-label, parallel, in pediatric patients (≤ 16) with fever ≥ 101°F (38.3°C).
Randomized to Caldolor 10 mg/kg vs Acetaminophen 10 mg/kg
- Stratified according to age (birth to < 6 months; 6 months to 16 years)
- PK measurements in Caldolor group collected
Tympanic route monitored every 15 min up to 105 min then at hours 2, 2.5, 3, 3.5, 4, & 6
Primary Outcome: Fever reduction within 2 hours after dose
Secondary Outcome: Fever reduction at 30, 60, and 240 minutes, and 4 and 24 hours; time to afebrile status and percentage of patients becoming afebrile after 4 hours.
N = 121 enrolled; 18 excluded; 103 patients received medication
- Caldolor = 47
- Acetaminophen = 53
Pediatric Pain & Fever Treatment52
Outcomes:
- IV Ibuprofen resulted in reduced temperatures at less than 4 hours and at 4 hours to 24 hours and less pain at 4 to 24 hours compared to acetaminophen.
- Adverse events were uncommon; acetaminophen and IV Ibuprofen appeared to have similar serious adverse event profiles.
Study description
- Meta-analysis of 19 studies with 241,138 participants, compared IV ibuprofen with acetaminophen.
- Primary outcomes were fever or pain within 4 hours of treatment onset. Safety outcomes included serious adverse events, kidney impairment, gastrointestinal bleeding, hepatotoxicity, severe soft tissue infection, empyema, and asthma and/or wheeze.
Demonstrated Fever Reduction in Adults1
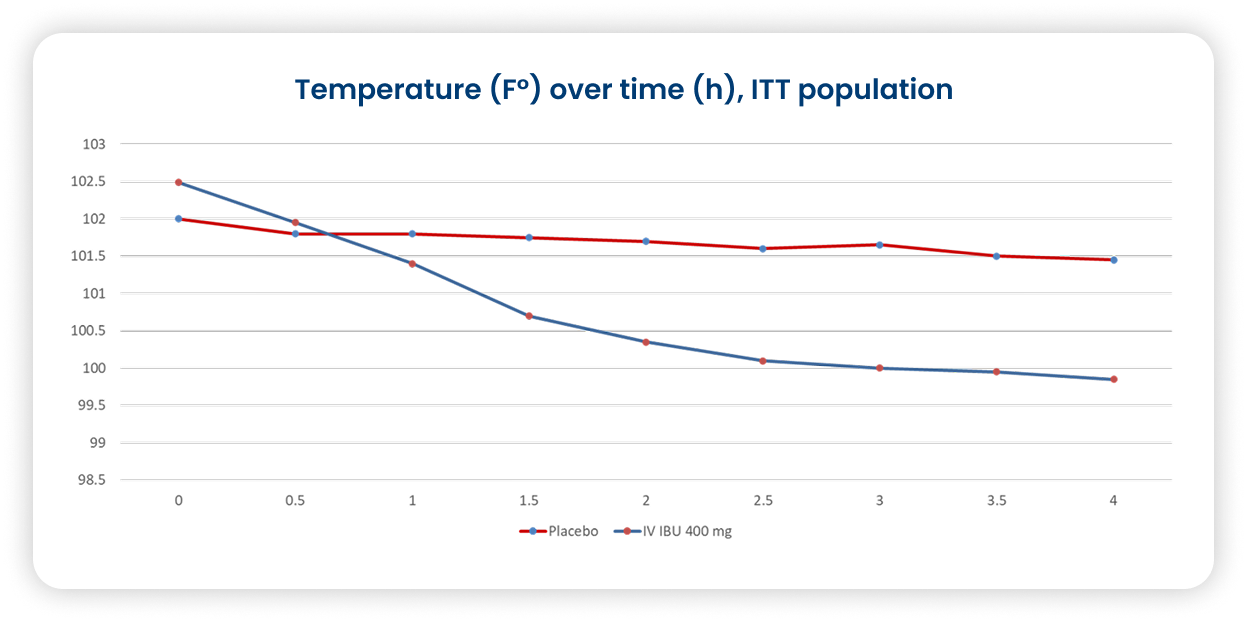
Caldolor 400mg effectively reduces fever at 4 hours in non-critically ill.53
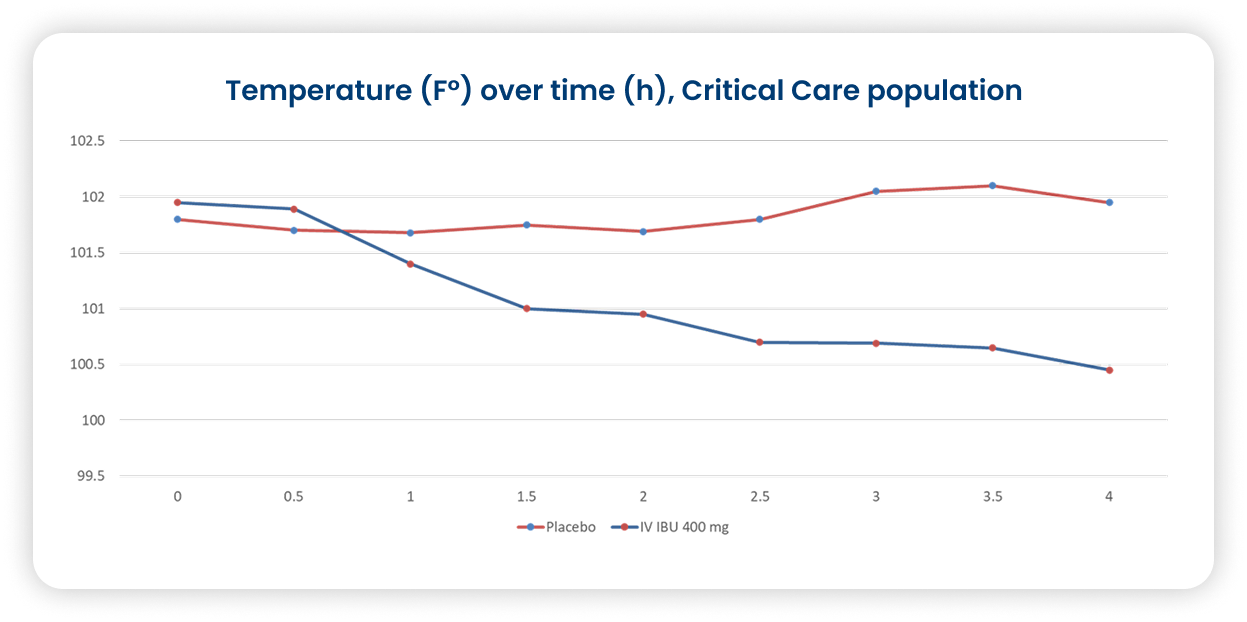
Caldolor 400mg effectively reduces fever at 4 hours in critically ill.53
Study description
- Multicenter, randomized, double-blind, parallel, placebo-controlled in patients with fever ≥ 101 °F
N = 120
- Placebo N = 28, 100 mg
- Caldolor N = 31, 200 mg
- Caldolor N = 30, 400 mg
- Caldolor N = 31
Primary outcome: percentage of subjects achieving a temperature <101.0 °F at 4 hours after initial dose
Secondary outcome: effect on temp at 24 hours
Fever Reduction in Burn Patients54
Caldolor significantly reduced fever in burn patients during the first 24 hours of treatment compared with placebo.
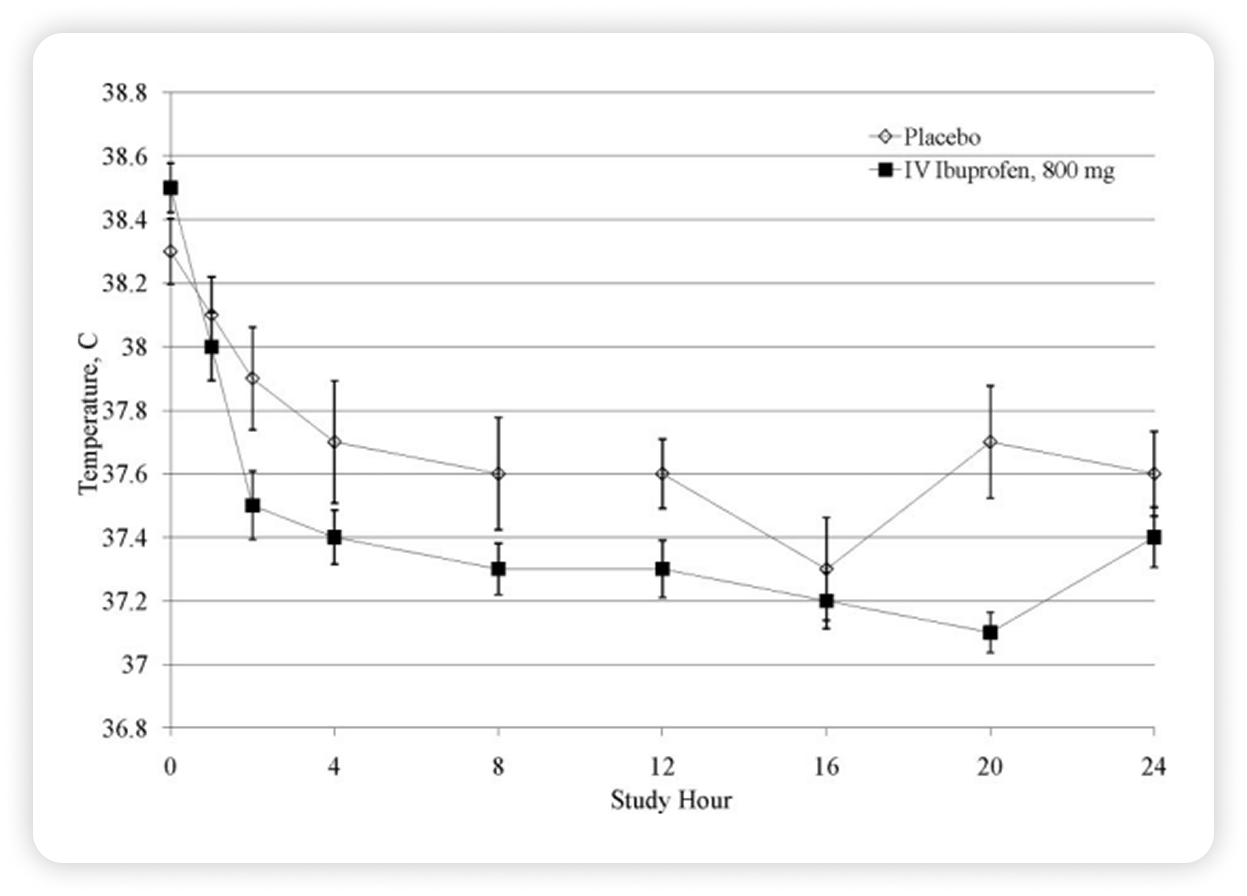
Over the full 120-hour dosing period, temperatures remained lower with Caldolor, and the treatment was generally well tolerated with no significant increase in serious adverse events compared with placebo.
Study description
- A prospective, multicenter, randomized, double-blind placebo-controlled trial of Caldolor for treatment of fever and pain in burn patients
- N = 61, randomly assigned in a 2:1 ratio to receive either 800 mg Caldolor or placebo every 6 hours for 120 hours
- Caldolor = 40
- Placebo = 21
- Primary outcome: area under the curve for temperature (AUC-T°) within the first 24 hours of treatment
Fever Reduction in Septic Patients45
Outcomes:
- Caldolor significantly reduced fever in septic patients, bringing temperatures back to normal, while fever remained elevated in the placebo group.
- Caldolor was more effective than acetaminophen for temperature control. As temperatures dropped in the ibuprofen group, acetaminophen use decreased; in the placebo group, acetaminophen use increased to 44 percent.
- Caldolor lowered physiologic markers associated with fever, including heart rate and metabolic demand, showing meaningful reductions in tachycardia, oxygen consumption, and lactate levels compared with placebo.
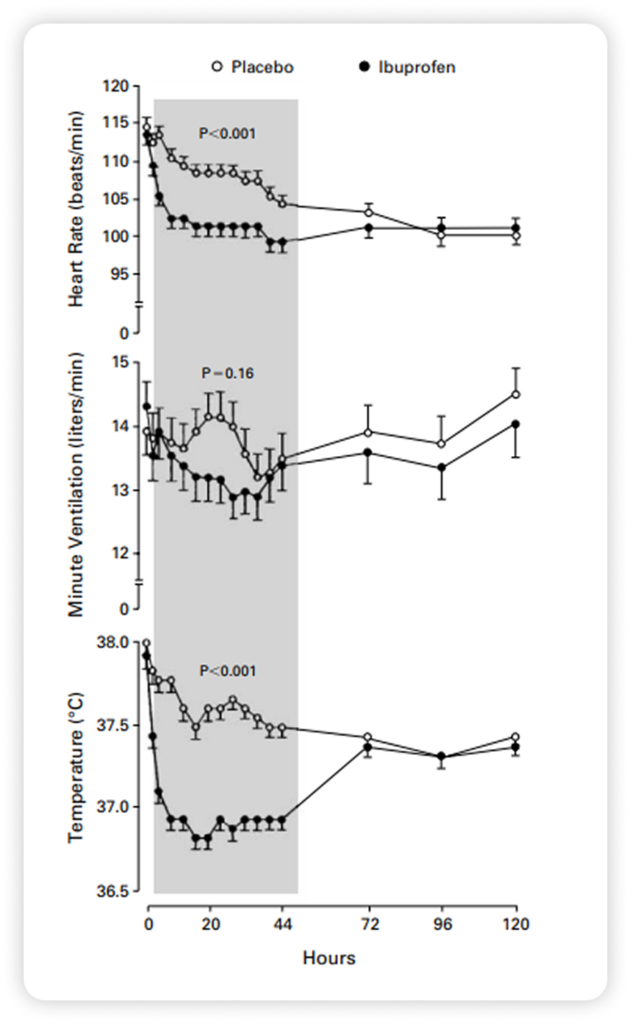
Study description
- A randomized, double-blind, placebo-controlled trial of intravenous Caldolor (10 mg per kilogram of body weight [maximal dose, 800 mg], given every six hours for eight doses) study of patients with sepsis, defined as fever, tachycardia, tachypnea, and acute failure of at least one organ system.
- N = 455
- Caldolor = 224
- Placebo = 231
Caldolor is a nonsteroidal anti-inflammatory drug indicated in adults and pediatric patients aged 3 months and older for the:
- Management of mild to moderate pain, including postoperative pain
- Management of moderate to severe pain, including postoperative pain, as an adjunct to opioid analgesics
- Reduction of fever
This Product Includes the Following Boxed Warning:
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events
Cardiovascular Thrombotic Events
- Non-steroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use.
- Caldolor is contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
Gastrointestinal Bleeding, Ulceration and Perforation
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events.
Important Dosage and Administration Instructions
- Caldolor Injection 800 mg/8 mL (100 mg/mL) vials must be diluted prior to administration.
- Caldolor Injection 800 mg/200 mL (4 mg/mL) bags are ready to use.
- Do not exceed 3,200 mg total daily dose in adults. Do not exceed 40 mg/kg or 2,400 mg, whichever is less, total daily dose in pediatric patients less than 17 years of age. In pediatric patients 3 months to less than 6 months of age, the dosage is limited to a single dose not to exceed 10 mg/kg or 100 mg, whichever is less.
Important Safety Information
Contraindications
- Caldolor is contraindicated in patients with known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to ibuprofen or any components of the drug product, and in patients who have a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients. Caldolor is contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
Warnings and Precautions
- Use with caution in patients with known cardiovascular (CV) disease or risk factors for CV disease, a history of peptic ulcer disease and/or GI bleeding, renal or liver disease or symptoms of hypertension, and heart failure. Use the lowest effective dose for the shortest time period in these patients to reduce the risk of serious adverse events. Avoid use in pregnant women starting at 30 weeks gestation.
Adverse Reactions
- The most common adverse reactions are nausea, flatulence, vomiting, headache, hemorrhage and dizziness (>5%). The most common adverse reactions in pediatric patients are infusion site pain, vomiting, nausea, anemia and headache (≥2%)
Have Questions or Would like to Stay Informed About Caldolor?
We’re Here to Help!
