Reduce Opioid Use with Caldolor
Caldolor Reducing Opioid Use in Surgical Settings
Caldolor Reducing Pain in Peds and Adults
Caldolor Reducing Fever in Peds and Adults
Caldolor’s Safety Profile
Pediatric Tonsillectomy3
Patients receiving Caldolor required 50% less doses of postoperative fentanyl in PACU, significantly less compared to placebo (P=0.021).
One of the most common pediatric procedures, tonsillectomy, may benefit from opioid reduction when Caldolor is used preoperatively.
This multicenter, randomized, double-blind placebo-controlled, single dose trial included 161 pediatric patients aged 6-17 years undergoing tonsillectomy. The primary endpoint was the number of doses and amount of postoperative fentanyl administered postoperatively for rescue analgesia.
Caldolor demonstrated a significant opioid-sparing effect in pediatric tonsillectomy patients.

Older Population15
This retrospective study aimed to evaluate the safety and efficacy of Caldolor in older patients. A post hoc subgroup analysis was performed with data from four multi-center, prospective clinical studies in which Caldolor was administered in patients 60 years of age or greater. Those who received Caldolor were administered an average total of 33.30 mg of morphine compared with 43.38 mg of morphine received by those in the placebo group, demonstrating a 23.2% reduction in total morphine consumption.
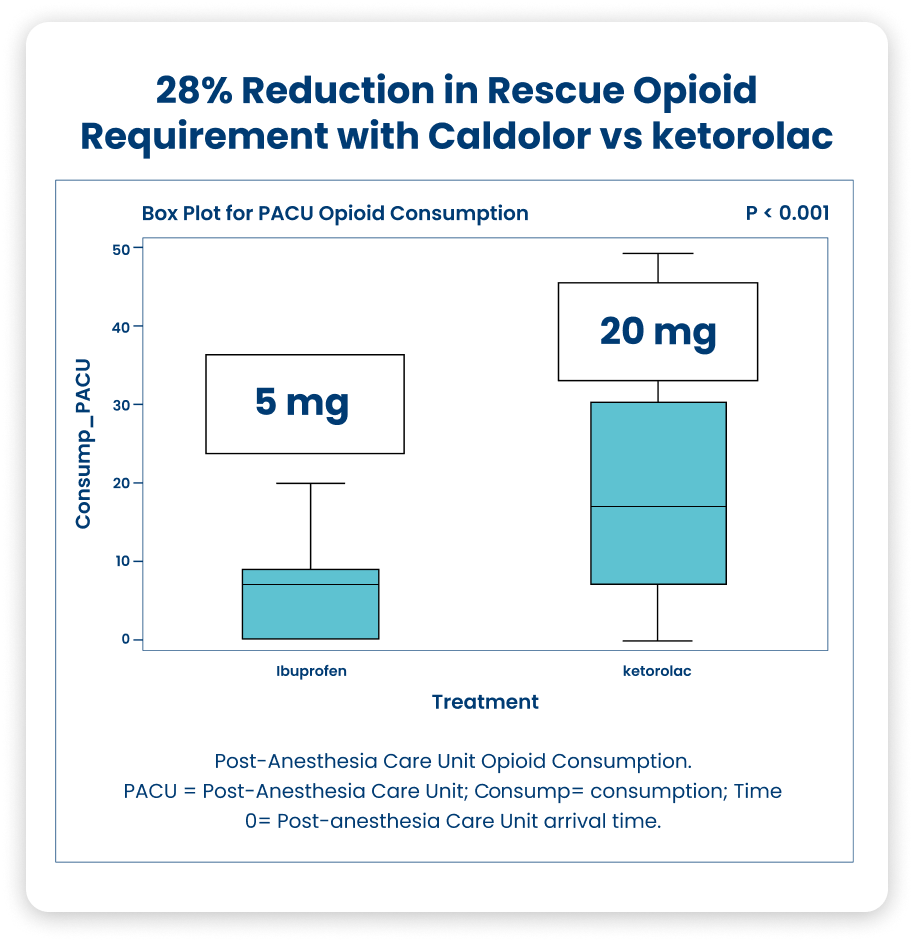
Arthroscopic Knee Surgery16
This single-center, randomized, double-blind, parallel, active comparator clinical pilot study was performed to assess the efficacy of two different analgesic treatment regimens: Caldolor and IV ketorolac, for the treatment of postoperative pain pertaining to arthroscopic knee surgery.

Abdominal Hysterectomy17
In this multi-center, randomized, double-blind, placebo-controlled trial, safety and efficacy of Caldolor as a postoperative analgesic following abdominal hysterectomy was evaluated. The morphine requirement was significantly reduced by 19% (P < 0.001) for those using Caldolor.

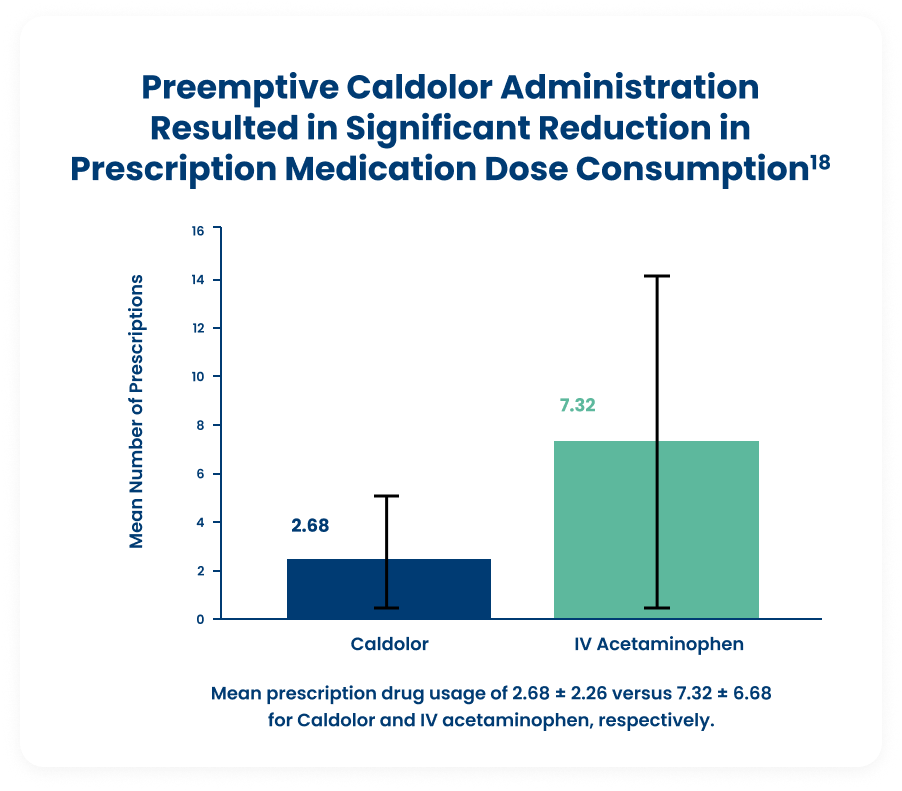
Oral Surgery — Third Molar Removal18
This prospective, randomized, single-blinded clinical trial, compared 800 mg of Caldolor and 1,000 mg of IV acetaminophen. The preemptive analgesic effect of Caldolor showed a significant reduction of prescription drug usage compared to IV acetaminophen (P = 0.005).
Orthopedic Trauma19
This single-center, randomized, double-blind, parallel-group, placebo-controlled trial evaluated the efficacy of Caldolor in the management of acute pain in orthopedic trauma patients and to minimize opioid use. In patients with moderate to severe orthopedic trauma, the initial dose of Caldolor (800 mg) reduced morphine requirements over 48 hours (P = 0.017), with reductions occurring as early as 8 hours. Caldolor showed a significantly longer pain relief time of 6.4 hours compared to the placebo pain relief time of 4.3 hours (P = 0.046).

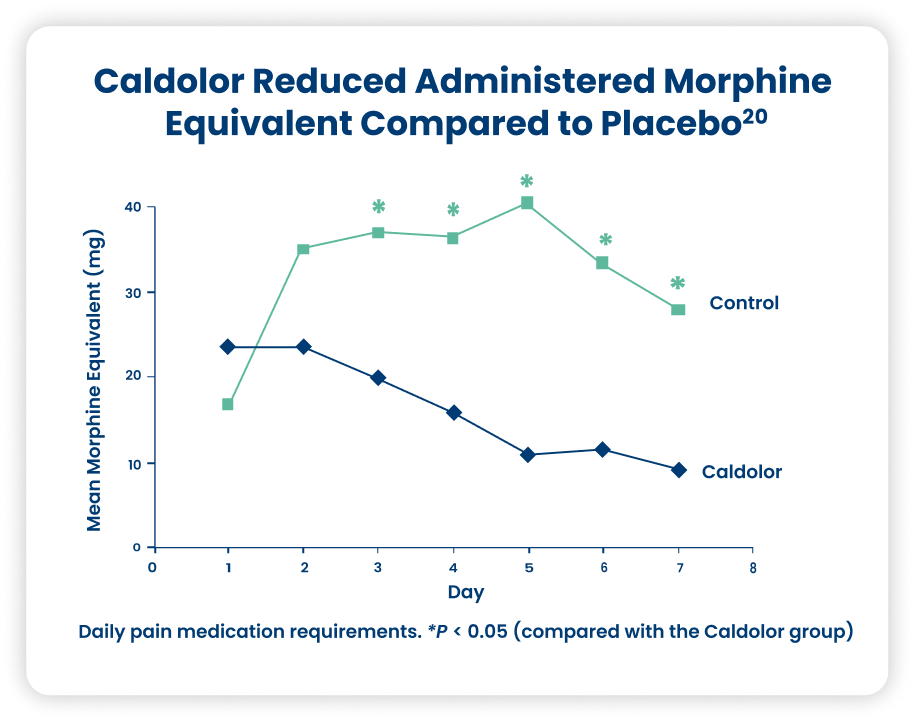
Rib Fracture20
This study is a retrospective chart review analyzing the pain management of patients 18 years of age or older who sustained rib fractures and were treated in a Level I trauma center. For those receiving Caldolor, the total weekly morphine-equivalent requirement was significantly decreased (P = 0.007). The daily morphine equivalent requirement did not differ between the study groups on Day 1 or Day 2; however, narcotic requirement was significantly decreased in Caldolor patients on each subsequent day of therapy (Days 3 through 7).
Transsphenoidal Surgery6
This single-center, randomized, double-blinded, placebo-controlled study involved adult patients with planned transsphenoidal surgery. Group 1 patients were treated with scheduled Caldolor, scheduled oral acetaminophen, and rescue opioids. Group 2 patients were treated with IV placebo, scheduled oral acetaminophen, and rescue opioids. Caldolor resulted in significantly decreased opioid use when compared to placebo.

Elective Orthopedic & Abdominal Surgery5
This multi-center, randomized, double-blind, placebo-controlled trial showed that there was a significant reduction in morphine requirements in patients that received intra-operative Caldolor vs placebo at 24 hours. A 22% reduction of morphine use was recorded in patients who were treated with Caldolor.

Elective Orthopedic2
In this multi-center, randomized, double-blind, placebo-controlled study with patients undergoing elective orthopedic surgery, patients in the placebo group required 59.5 mg of morphine during the 28-hour observation period while patients treated with Caldolor required 41.1 mg of morphine. Thus in a setting where patients had equal access, patients who received Caldolor used 30.9% less morphine (P < 0.001) than those receiving placebo.

Caldolor is a nonsteroidal anti-inflammatory drug indicated in adults and pediatric patients aged 3 months and older for the:
- Management of mild to moderate pain, including postoperative pain
- Management of moderate to severe pain, including postoperative pain, as an adjunct to opioid analgesics
- Reduction of fever
This Product Includes the Following Boxed Warning:
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events
Cardiovascular Thrombotic Events
- Non-steroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use.
- Caldolor is contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
Gastrointestinal Bleeding, Ulceration and Perforation
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events.
Important Dosage and Administration Instructions
- Caldolor Injection 800 mg/8 mL (100 mg/mL) vials must be diluted prior to administration.
- Caldolor Injection 800 mg/200 mL (4 mg/mL) bags are ready to use.
- Do not exceed 3,200 mg total daily dose in adults. Do not exceed 40 mg/kg or 2,400 mg, whichever is less, total daily dose in pediatric patients less than 17 years of age. In pediatric patients 3 months to less than 6 months of age, the dosage is limited to a single dose not to exceed 10 mg/kg or 100 mg, whichever is less.
Important Safety Information
Contraindications
- Caldolor is contraindicated in patients with known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to ibuprofen or any components of the drug product, and in patients who have a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients. Caldolor is contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
Warnings and Precautions
- Use with caution in patients with known cardiovascular (CV) disease or risk factors for CV disease, a history of peptic ulcer disease and/or GI bleeding, renal or liver disease or symptoms of hypertension, and heart failure. Use the lowest effective dose for the shortest time period in these patients to reduce the risk of serious adverse events. Avoid use in pregnant women starting at 30 weeks gestation.
Adverse Reactions
- The most common adverse reactions are nausea, flatulence, vomiting, headache, hemorrhage and dizziness (>5%). The most common adverse reactions in pediatric patients are infusion site pain, vomiting, nausea, anemia and headache (≥2%)
Have Questions or Would like to Stay Informed About Caldolor?
We’re Here to Help!
