Confidence Backed by Clinical Evidence
Caldolor has a well-established safety profile in pediatrics, adults, and the older adult populations.3,36,44
Adverse Events
An integrated safety analysis of 10 clinical studies involving Caldolor in adults demonstrated that Caldolor can be safely administered prior to surgery and continued in the postoperative period as a component of multimodal pain management.36
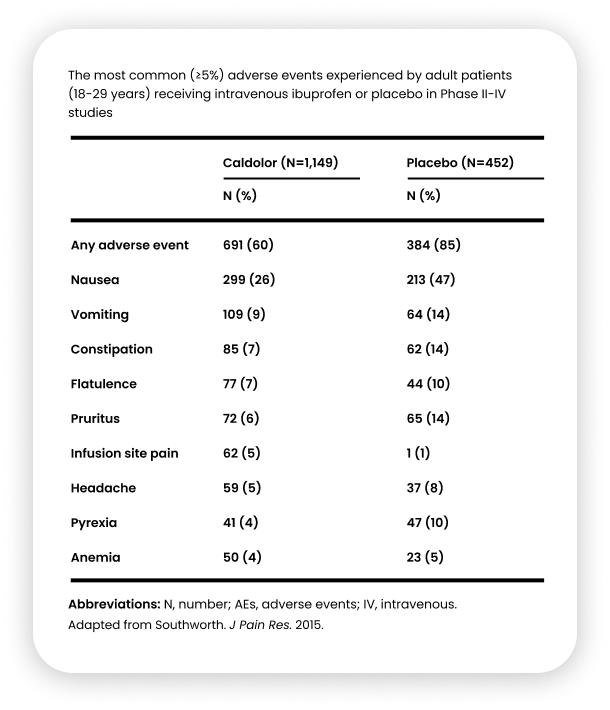
Caldolor demonstrated a strong safety profile, with a smaller percentage of adverse events (60%) compared with placebo (85%).36
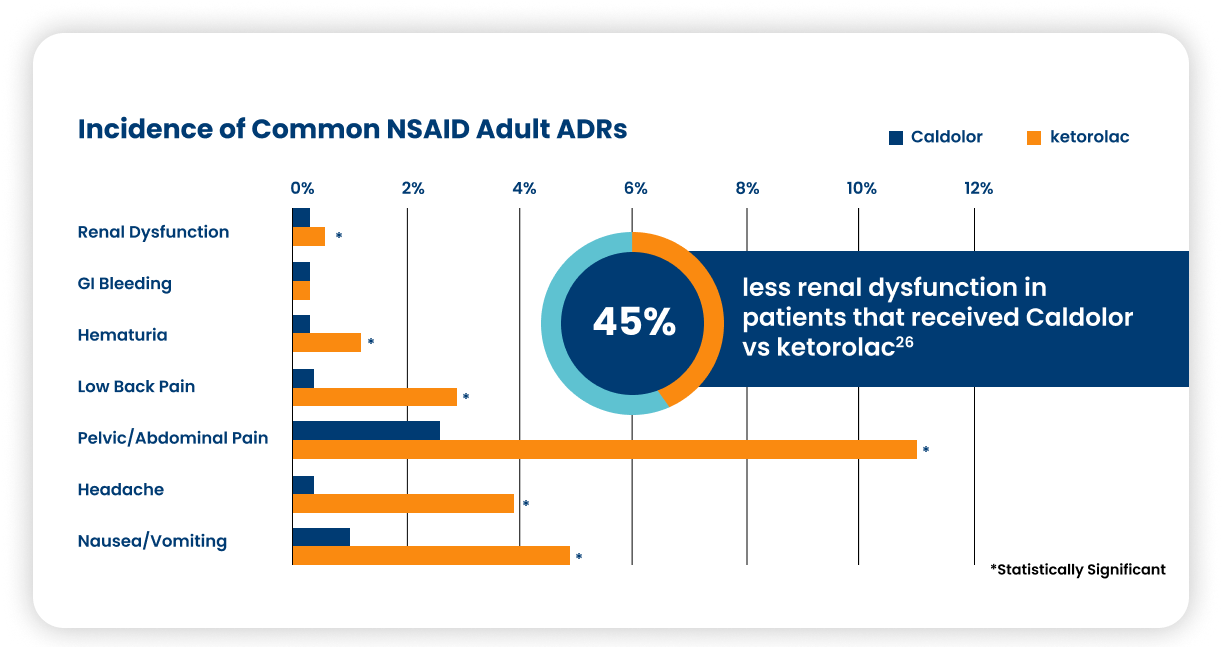
A real-world evaluation of Caldolor to ketorolac shows Caldolor is associated with fewer adverse drug reactions in adults.26
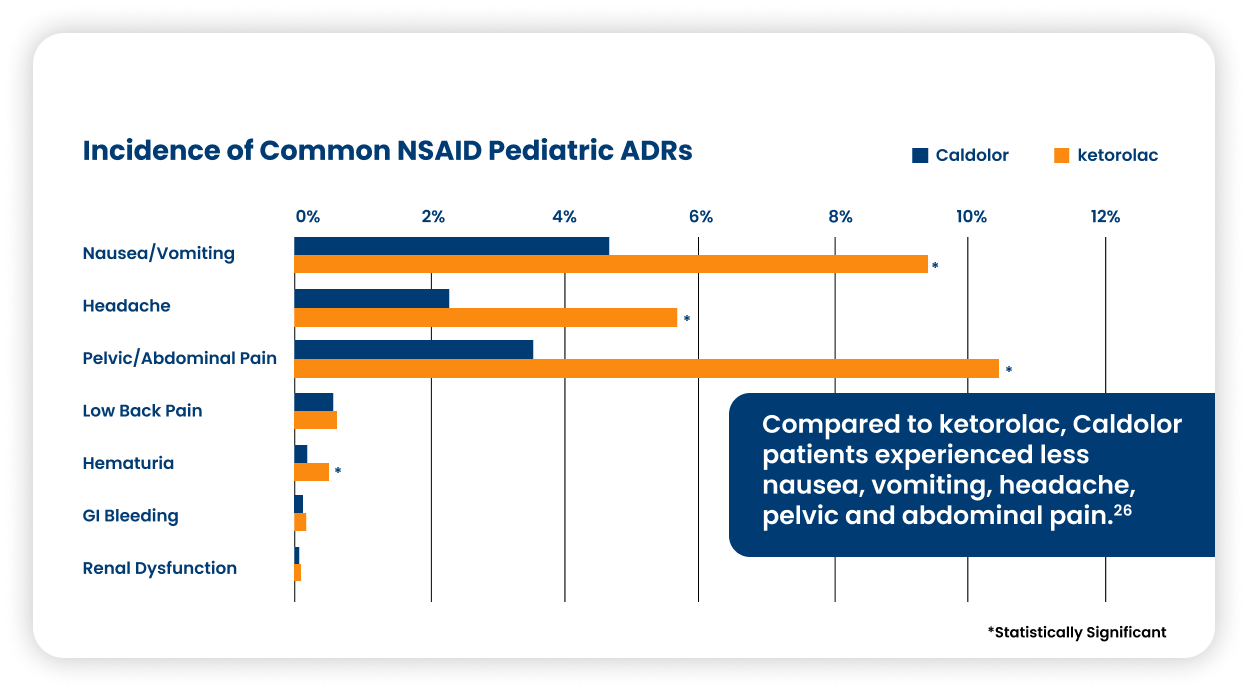
A real-world evaluation shows Caldolor is associated with fewer adverse reactions in pediatrics vs ketorolac.26
Most Common Adverse Reactions
The most common adverse reactions are nausea, flatulence, vomiting, headache, hemorrhage and dizziness (>5%).1
The most common adverse reactions in pediatric patients are infusion site pain, vomiting, nausea, anemia, and headache (≥2%).1
To report SUSPECTED ADVERSE REACTIONS, contact Cumberland Pharmaceuticals Inc. at 615-425-7642 or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
Pediatric Use
Caldolor is well tolerated in pediatric patients older than 1 month and younger than 6 months.44
Single and multiple 10 mg/kg doses of Caldolor trended safe, with no renal concerns. Drug exposure following a single dose of Caldolor in infants 1 to 6 months of age was similar to what was previously reported in older children.
Caldolor showed a safe and well-tolerated profile in pediatric tonsillectomy patients.3
There were no significant differences in the incidence of serious adverse events, surgical blood loss (P = 0.662), incidence of postoperative bleeding, or a need for surgical re-exploration between the treatment groups.
Older Population Use
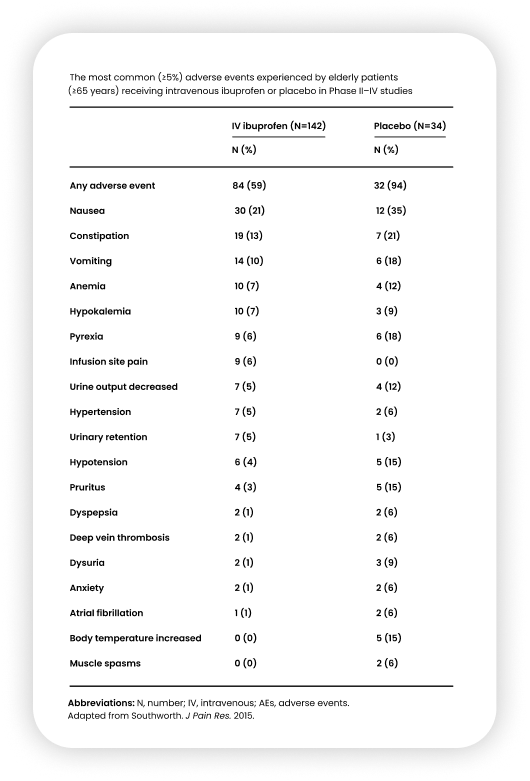
A post-hoc subgroup analysis evaluated Caldolor in patients aged 60 years and older undergoing elective surgery. Safety outcomes were favorable with AEs occurring 55% of Caldolor-treated patients vs 90% with placebo, while serious AEs were similar between groups.15Importantly, there were no increases in gastrointestinal, renal, cardiovascular, or bleeding complications in the Caldolor group.
An integrated safety analysis of 10 clinical studies involving Caldolor in adults demonstrated that Caldolor can be safely administered prior to surgery and continued in the postoperative period as a component of multimodal pain management. A subset analysis of elderly patients suggests that no dose adjustment is needed in this higher-risk population.36
Elderly patients (≥ 65 yo) experienced fewer total AEs (59% vs 94%).
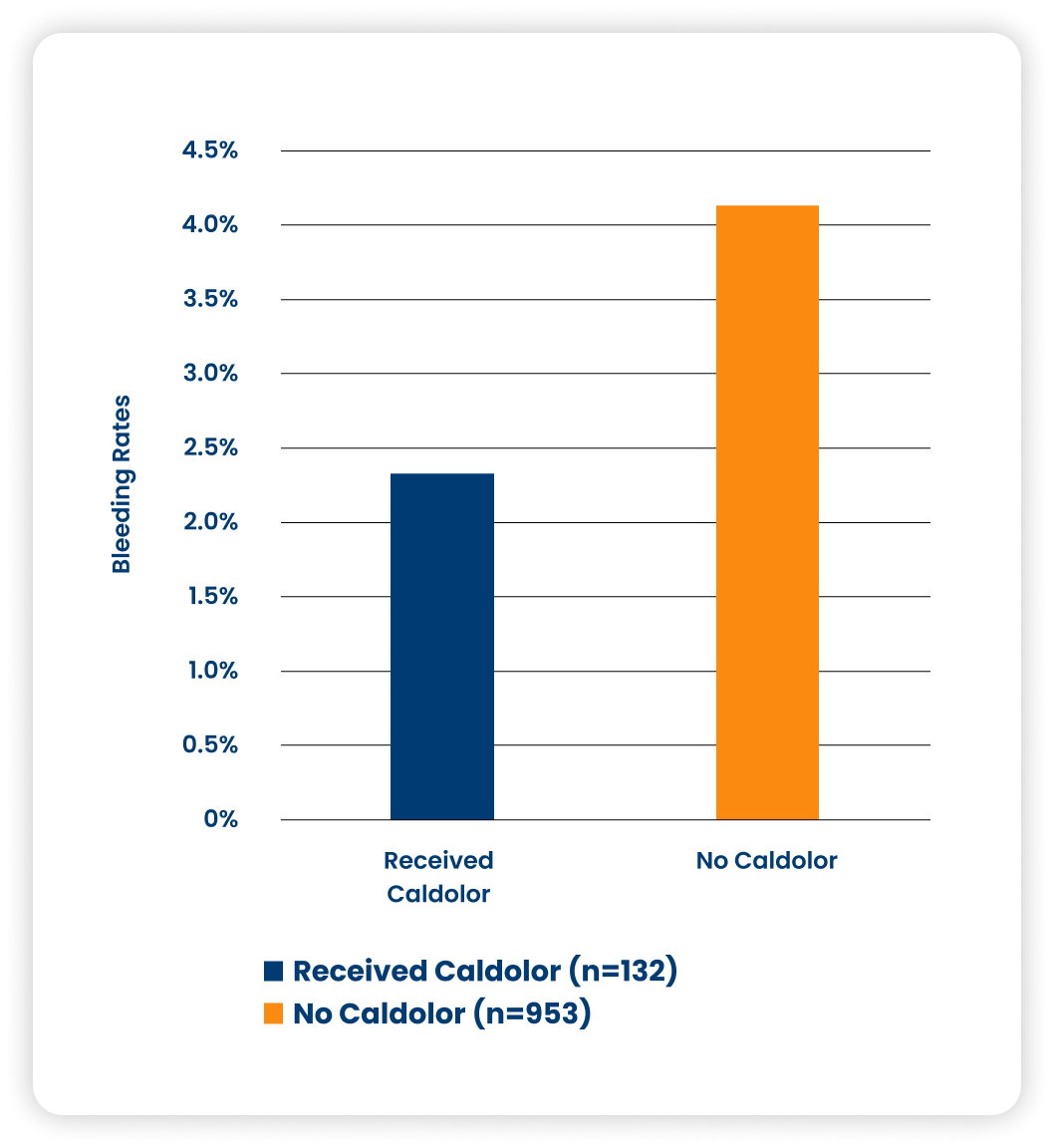
Bleeding
In a prospective, phase 3, randomized, placebo-controlled, double-blind study of patients with severe sepsis syndrome, the results demonstrated no differences in bleeding events, transfusion requirements, or coagulation abnormalities between Caldolor and placebo-treated patients at any time point in the study.45
Pediatric patients undergoing tonsillectomy showed no difference in primary bleeding rate or secondary bleeding rate between the Caldolor and placebo-treated patients. Caldolor use is not associated with increased post-tonsillectomy bleeding.4
An additional study evaluated Caldolor added to oral acetaminophen in patients undergoing transsphenoidal surgery, and no bleeding complications from the study were related to the study drug as determined by the data review committee.6
Bone Healing
Prostaglandins play a key regulatory role in osteoblast and osteoclast function; however, because the bone remodeling phase does not typically begin until approximately 5–16 days after orthopedic injury, acute NSAID administration is unlikely to impair bone healing.46,47
Human clinical data further demonstrated that short-term, therapeutic-dose NSAID use does not adversely affect bone healing.48,49 Meta-analytic evidence suggests that reported associations between NSAID exposure and delayed union or nonunion are largely driven by patient age, an established independent risk factor for impaired fracture healing.50
Prospective data strengthen these observations. In a randomized, blinded trial of pediatric patients with long-bone fractures, ibuprofen administered for acute fracture-related pain compared to acetaminophen did not impair clinical or radiographic healing in skeletally immature patients.49
Caldolor is a nonsteroidal anti-inflammatory drug indicated in adults and pediatric patients aged 3 months and older for the:
- Management of mild to moderate pain, including postoperative pain
- Management of moderate to severe pain, including postoperative pain, as an adjunct to opioid analgesics
- Reduction of fever
This Product Includes the Following Boxed Warning:
Warning: Risk of Serious Cardiovascular and Gastrointestinal Events
Cardiovascular Thrombotic Events
- Non-steroidal anti-inflammatory drugs (NSAIDs) cause an increased risk of serious cardiovascular thrombotic events, including myocardial infarction and stroke, which can be fatal. This risk may occur early in treatment and may increase with duration of use.
- Caldolor is contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
Gastrointestinal Bleeding, Ulceration and Perforation
- NSAIDs cause an increased risk of serious gastrointestinal (GI) adverse events including bleeding, ulceration, and perforation of the stomach or intestines, which can be fatal. These events can occur at any time during use and without warning symptoms. Elderly patients and patients with a prior history of peptic ulcer disease and/or GI bleeding are at greater risk for serious GI events.
Important Dosage and Administration Instructions
- Caldolor Injection 800 mg/8 mL (100 mg/mL) vials must be diluted prior to administration.
- Caldolor Injection 800 mg/200 mL (4 mg/mL) bags are ready to use.
- Do not exceed 3,200 mg total daily dose in adults. Do not exceed 40 mg/kg or 2,400 mg, whichever is less, total daily dose in pediatric patients less than 17 years of age. In pediatric patients 3 months to less than 6 months of age, the dosage is limited to a single dose not to exceed 10 mg/kg or 100 mg, whichever is less.
Important Safety Information
Contraindications
- Caldolor is contraindicated in patients with known hypersensitivity (e.g., anaphylactic reactions and serious skin reactions) to ibuprofen or any components of the drug product, and in patients who have a history of asthma, urticaria, or other allergic-type reactions after taking aspirin or other NSAIDs. Severe, sometimes fatal, anaphylactic reactions to NSAIDs have been reported in such patients. Caldolor is contraindicated in the setting of coronary artery bypass graft (CABG) surgery.
Warnings and Precautions
- Use with caution in patients with known cardiovascular (CV) disease or risk factors for CV disease, a history of peptic ulcer disease and/or GI bleeding, renal or liver disease or symptoms of hypertension, and heart failure. Use the lowest effective dose for the shortest time period in these patients to reduce the risk of serious adverse events. Avoid use in pregnant women starting at 30 weeks gestation.
Adverse Reactions
- The most common adverse reactions are nausea, flatulence, vomiting, headache, hemorrhage and dizziness (>5%). The most common adverse reactions in pediatric patients are infusion site pain, vomiting, nausea, anemia and headache (≥2%).
Have Questions or Would like to Stay Informed About Caldolor?
We’re Here to Help!
